Our Technology

The Need
The gold standard of mitral valve
replacement surgery is indicated for only 20% of MR patients,
resulting in only 30,000 surgeries, while conservative treatment with medication is not sufficient.
Mitral Regurgitation (MR) is the most frequent valve disease in the United States.
Over 7,000,000 people in the U.S. suffer from MR,
representing almost 2% of the nation’s population, and 10% of the population above 75 years.
There is an increasing number of patients with mitral valve disorders, with over
250,000 new patients suffering of mitral regurgitation disease each year.
What is Mitral Regurgitation
Mitral Regurgitation (MR) has multiple mechanisms and is divided into Degenerative or Primary MR and Functional or Secondary MR. Surgical Mitral Valve Replacement is the gold standard for treatment of MR, with excellent clinical outcomes; however, it is inappropriate for most patients, due to their underlying clinical condition and the risks associated with the surgery procedure (open-heart CPB surgery). Only 30,000 patients undergo such replacement surgery in the US each year, out of 4M moderate to severe MR patients who could benefit from treatment. In addition to replacement surgery, repair techniques, either minimally invasive or via surgery, also exist, but are considered to offer incomplete solutions for most patients. Hence, the apparent need for a minimally-invasive, catheterization-based procedure, to perform replacement of the mitral valve (TMVR – Transcatheter Mitral Valve Replacement). At Innovalve, we have developed a novel catheter-based technological approach, applicative to multiple heart valve morbidities. Our solution utilizes a unique technological solution, patent-protected, based on specially designed arms, which via a rotation maneuver both allows robust anchoring of an implant to the native tissue, as well manipulation of the natural orifice to fit the prosthesis.
The main challenge
The main challenge of any Transcatheter Mitral Valve Replacement (TMVR) technology is to secure robust anchoring of the valve prosthesis to the mitral annulus while achieving adequate sealing. To utilize a transseptal catheterization approach, the prosthesis must be small enough, so it has a short and narrow encapsulation profile.

Innovalve’s breakthrough technology has been used to develop our first product – a minimally-invasive, catheterization-based, Transcatheter Mitral Valve Replacement (TMVR) System – Innostay.
Innovalve’s Innostay has successfully completed its development phase, as well as rigorous pre-clinical testing, including chronic and acute animal studies, benchtop model simulations, biocompatibility, and sterilization. Innovalve’s TMVR received FDA approval to commence limited clinical trials in the US, under the IDE-EFS pathway (Investigational Device Exemption – Early Feasibility Study). Innovalve also received Israeli MOH clearance for limited clinical studies and is in process with other European territories. Innovalve plans to commence its clinical trial activities during the 2nd half of 2021.
Advantages of Innovalve’s TMVR Innostay System

Robust anchoring and excellent sealing using a patented rotation maneuver
with a circumferential arm design

As the Innostay TMVR brings the native tissue to the device, it is
indifferent to the shape and size of the mitral valve

The small,
crimped device enables transseptal delivery,
easy deployment, and a quick procedure

OUR TMVR INNOSTAY SYSTEM IS COMPRISED OF TWO MAIN COMPONENTS
Valve Prosthesis
The Valve Prosthesis (patented) is a permanent implant fabricated from bovine pericardial tissue leaflets mounted onto a self-expanding nitinol metal alloy frame.
Delivery System
The delivery system is designed to enable safe and accurate deployment of the valve prosthesis in the mitral annulus and utilizes the more desired minimally invasive transfemoral-transseptal approach. Innovalve’s delivery system (patent pending) employs a 5-catheter and 3-handle configuration.
Intended Use
The Innovalve Transcatheter Mitral Valve Replacement (TMVR) system is intended for use in adult patients with symptomatic moderate-severe and severe mitral regurgitation who are at elevated risk for surgical mitral valve repair or replacement and who are anatomically eligible for a transfemoral catheterization with transseptal approach to the left atrium.
How it works
The Innovalve transseptal delivery system is designed to enable safe deployment of the Innovalve implant. The delivery system accesses the vascular anatomy at the femoral vein, and advances over a guidewire through the inferior vena cava into the right atrium of the heart. A septal crossing is performed to reach the left atrium and the mitral valve. The steerable catheter delivery system provides direction and orientation to position the capsule containing the prosthesis valve at the mitral annulus. Once inside the mitral space, the implant is deployed in a controlled fashion, rotated to grasp the chordae, positioned and anchored properly, and then released from the delivery system.
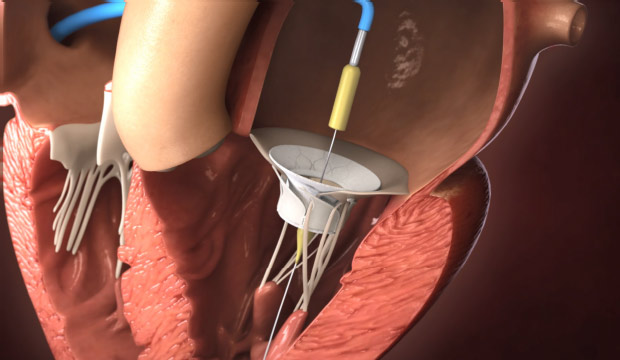
Step 1
Delivery of device to mitral orifice
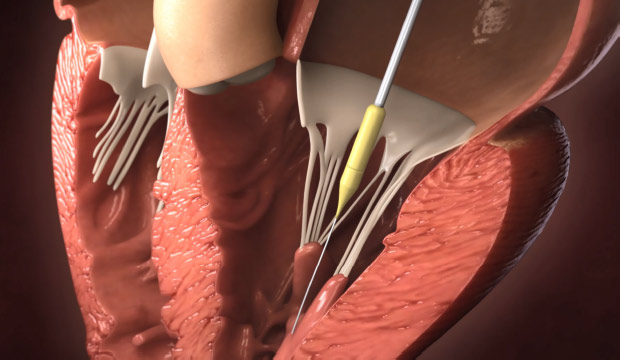
Step 2
Delivery of device to mitral orifice
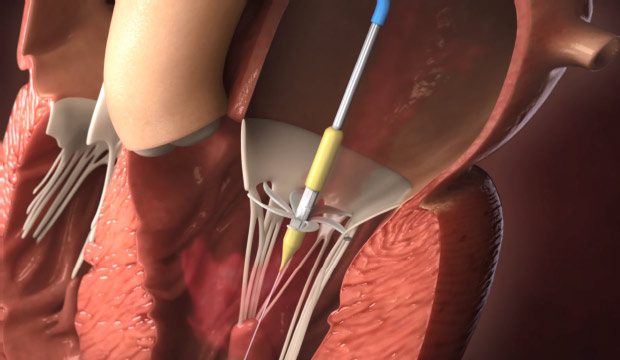
Step 3
Rotation of the device arms and capturing of the chordae
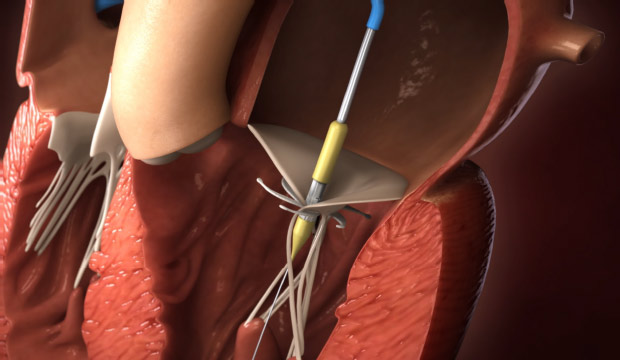
Step 4
Deployment of the prosthetic valve and release of the valve from the delivery system